Meniscal Transplant Surgery
CMI (Collagen based Biosynthetic Scaffold)

The Collagen Meniscus Implant (CMI®) is a biological and completely absorbable implant made from highly purified collagen with a porous structure.
It is arthroscopically attached to fill the void resulting from damaged and lost meniscal tissue and makes use of the body's own healing ability.
The porous implant matrix initially serves as a guide for the migration of the body's own cells and is completely absorbed afterwards. More than 16 years of clinical experience has proven that this is an established procedure for meniscus treatment. A defect filled with new tissue potentially reduces the risk of arthritis and produces outstanding clinical results, thus improving patients' quality of life and physical resilience.
CMI is intended for use in patients with an irreparable meniscus tear or loss of meniscus tissue.
When a meniscus injury can't be reattached by suturing, it is considered an 'irreparable meniscus tear'. Often the defective meniscal parts must be removed. If parts of the meniscus are removed without substitution, the upper and lower leg rub directly on each other and the cartilage layer is gradually worn down.
Studies have revealed that even partial removal of the meniscal tissue can increase cartilage stresses and lead to degenerative changes of the articular surfaces. These can result in considerable pain and loss of motion. Due to the degenerative nature of osteoarthritis, patients may eventually need an artificial knee joint.
The Collagen Meniscus Implant was developed to prevent or delay these long-term consequences of partial meniscectomy.
Once it's determined a patient is a good candidate for the CMI procedure, the steps are fairly simple. The suitability is dependent on how much meniscal tissue is left and how much wear and damage is already present in the knee.
A brief overview of what the patient can expect is outlined below:
- The surgeon will perform a routine diagnostic arthroscopy to confirm the meniscus injury and the appropriateness of the CMI procedure.
- The surgeon will prepare the meniscus by removing any damaged tissue.
- The meniscus defect will be measured, and the CMI scaffold will be trimmed to fit.
- The CMI scaffold will be placed into the knee joint through an arthroscopic portal.
- The CMI device will be sutured in place.
- The surgeon will close the small openings in the knee that were used to access the joint space.
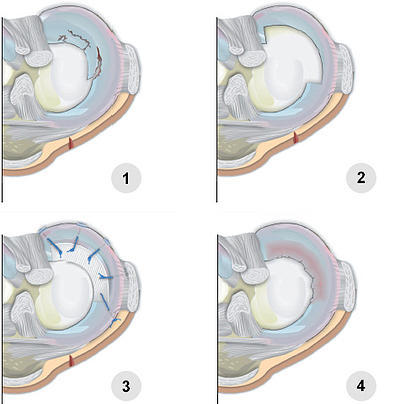
CMI Implantation image (1-4pics)
- Irreparable meniscus tear
- Resected meniscus damage / prepared implant site
- Implanted CMI
- New meniscus-like tissue
Rehabilitation
Patients who have undergone CMI implantation often feel able to return to their usual activities sooner than they had expected. Therefore, it is vital that the patient remind themselves, that although the operated knee may not be causing discomfort, the actual healing process has only just begun.
Download the full rehabilitation protocol here: click here for more information about Rehabilitation Protocol
Published results with the collagen based biosynthetic scaffold have shown improved pain and function levels at 10 years follow up and it potentially protects their knee from further damage and reduces the onset of arthritis.
Allograft
When patients have had an extensive meniscal resection in the past, due to a large tear, they may not have much meniscal tissue left and in this group of patients it is difficult to suture a biosynthetic graft. It may be more suitable for this group of patients to have their whole meniscus replaced using an allograft.
Tissue surgically transplanted from one person to another is called allograft. Bone, tendons and cartilage, skin, heart valves and veins are common types of tissues that are used for transplant to help patients in many different types of surgeries.
The most common allograft procedure in the UK is ACL reconstruction where non-bone tendons are routinely used to replace damaged ligaments in the knee.
Every surgical procedure involves risk from multiple factors. The question is how to reduce that risk to an absolute minimum.
The safety of any tissue is contingent upon three stages – donor screening, laboratory testing and tissue preparation validated to address potential disease transmission.
Screening, Testing and Sterilising for Patient Safety:
A complete medical/social history must be performed for every donor including cause of death, lifestyle risk assessment and family interviews.
Beyond donor screening, an extensive panel of serological and microbiological tests are performed.
These results are subject to stringent acceptance criteria in order to release the donor tissue.
In addition to serological testing on the donor's blood, microbiological testing is used throughout the process (where appropriate) to screen for potential contamination and to provide confirmation of tissue suitability for transplant.
All soft tissue allografts provided by Hospital Innovations are sterilised through a patented process called Biocleanse.
The BioCleanse system sterilizes tissue using a complex, proprietary combination of mechanical and chemical processes, working in conjunction with each other. The BioCleanse process does not sterilize using excessive heat or irradiation so the tissue benefits from the highest levels of sterility, whilst maintaining its mechanical strength.
Overall published results show that 70-75% of patients have improved pain and function levels following the allograft meniscal transplant procedure and it potentially protects their knee from further damage and reduces the onset of arthritis.
